|
When an electron is added to such a small atom, increased electron–electron repulsions tend to destabilize the anion. This is due to the structural similarities’ elements have within a period or family that allows for these. 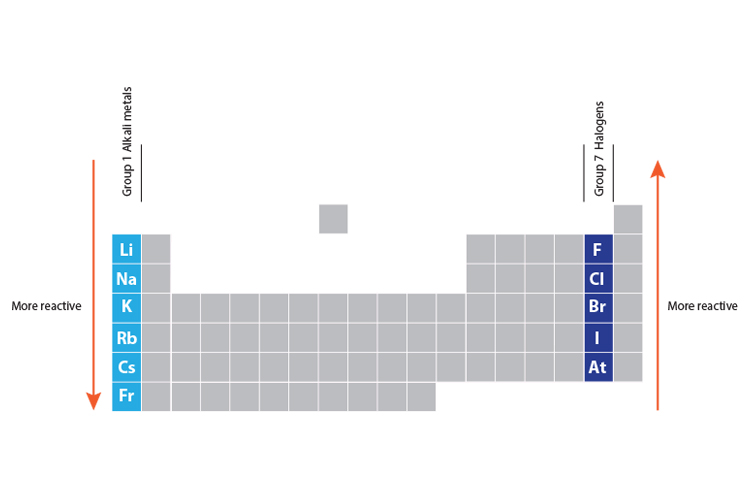
These trends have allowed scientists in the past to predict certain characteristics of unknown elements. In contrast to the chemistry of the second-period elements, the chemistry of the third-period elements is more representative of the chemistry of the respective group.ĭue to their small radii, second-period elements have electron affinities that are less negative than would be predicted from general periodic trends. Periodic trends are observable patterns in the properties of an element that are dependent on its position in the Periodic Table. The anomalous chemistry of second-period elements results from three important characteristics: small radii, energetically unavailable d orbitals, and a tendency to form pi (π) bonds with other atoms. In chemistry, periodic trends are specific patterns that are present in the periodic table that illustrate different aspects of certain elements when. Consequently, the elements of the third period (n = 3: Na, Mg, Al, Si, P, S, and Cl) are generally more representative of the group to which they belong. The chemistry of the second-period element of each group (n = 2: Li, Be, B, C, N, O, and F) differs in many important respects from that of the heavier members, or congeners, of the group. Unique Chemistry of the Lightest Elements The semimetals lie along the diagonal line separating the metals from the nonmetals and exhibit intermediate properties. Consequently, the elements in the upper right of the periodic table are the smallest and most electronegative the elements in the bottom left are the largest and least electronegative. In contrast, atomic size decreases from left to right and from bottom to top. 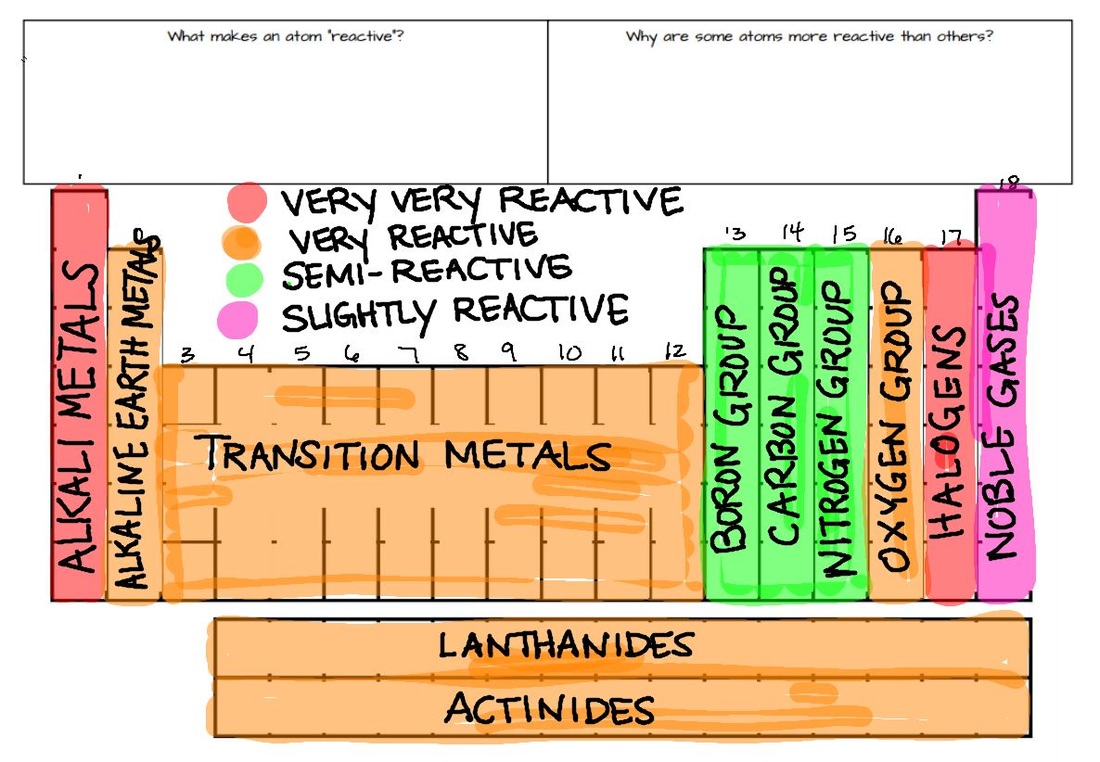
Part of Chemistry (Single Science) Structures, trends, chemical. Ionization energies, the magnitude of electron affinities, and electronegativities generally increase from left to right and from bottom to top. The periodic table helps to categorise the known elements and make predictions about ones that we haven’t yet discovered. 
\): Summary of Periodic Trends in Atomic Properties.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
